Mammut hunters and their African origin
Posted by Sjur Cappelen Papazian on March 9, 2016
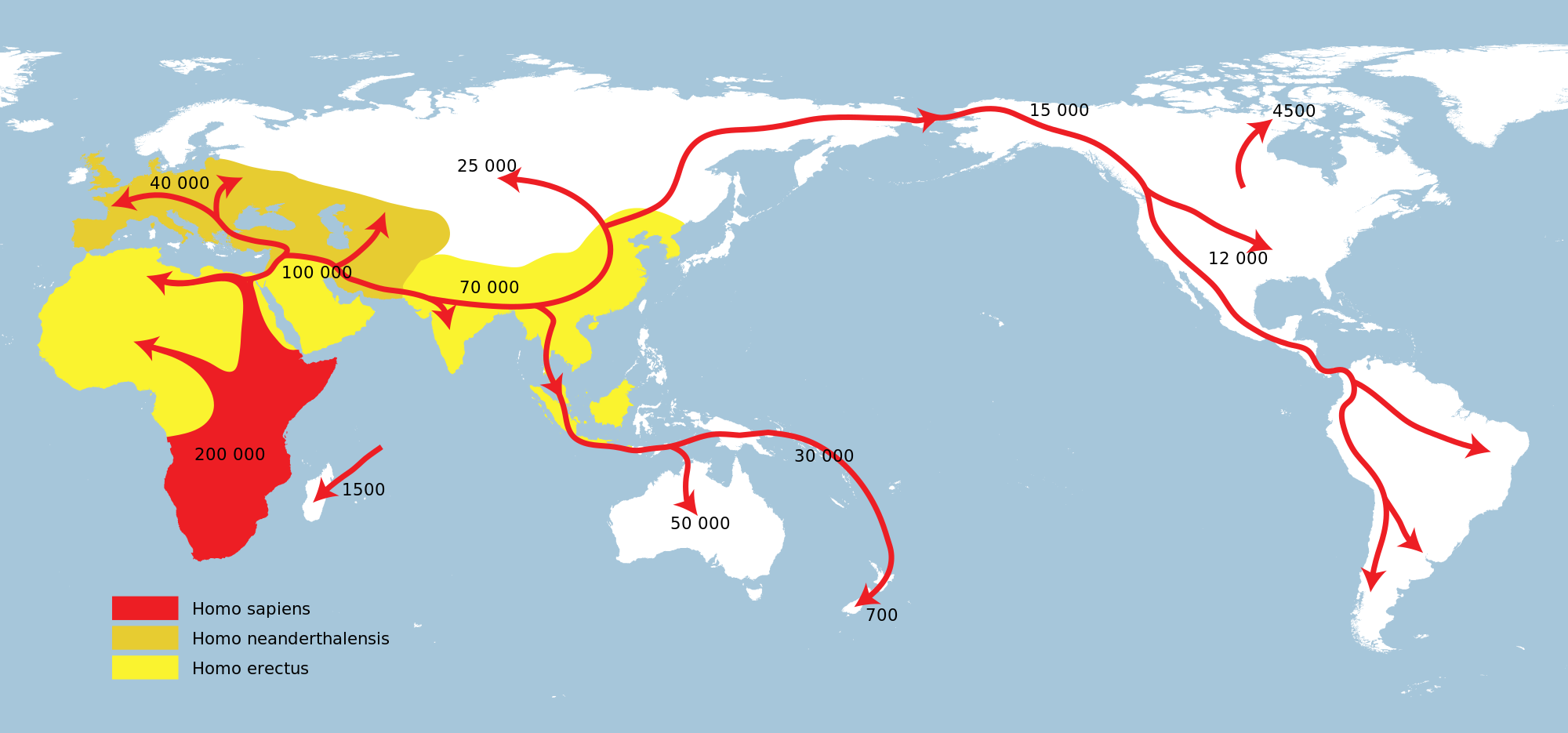
Human Expansion

Mammut Hunters
Haplogroup R

Haplogroup R1b
Haplogroup E

Haplogroup E-V38

Bantu Expansion

Bantu

Khoisan
The recent origin for most paternal lineages in west Central Africa is a result of the Bantu expansion starting ∼5,000 years ago, having erased virtually all previous Y-chromosome diversity of populations inhabiting this region.
The nonnegligible presence of the R1b1* lineage in west Central African samples (with a frequency over 5%) might point toward additional demographic expansions within the area besides the “Bantu expansion.”
The geographic origin of the R1b lineage is situated Eurasia and not in Africa. Its sporadic presence, although at low frequencies, in some African populations has been proposed to result from back migrations from Eurasia into Africa during ancient times.
The internal STR diversity of this lineage in west Central Africa points toward a putative expansion occurring 7,000 years ago, before the “Bantu expansion”, having been shown to be especially frequent in northern Cameroon, near the putative Bantu expansion origin.
While Western Europe is dominated by the R1b1a2 (R-M269) branch of R1b, the mostly Chadic-speaking area in Africa is dominated by the branch known as R1b1c (R-V88). These represent two very successful twigs on a much bigger family tree.
The R1b1 is result of a mid-Holocene migration of proto-Chadic Afroasiatic speakers through the Central Sahara into the Lake Chad Basin, and its presence in North Africa may be explained as the result of a migration from the south.
R1b1a is not directly tied to Proto-Afro-Asiatic. Proto-Aafro-Asiatic might already have been spoken in Northeast Africa and the Horn by E1b1b hunter gatherers when R1b1 farmers arrived on the scene, but the R1b1 carriers adopted some proto form of Afro-Aasiatic after settling in Africa.
R1b1 carriers made it’s way to Africa with pastoralism, or farming and cattles, since all African R1b1 populations have people that are positive for the 13910 C/T polymorphism, the SNP that allows lactase persistence in adulthood in North Indian and European populations and spread into Europe during the Neolithic, which has been estimated to have first appeared in humans 5,000 to 12,000 years ago.
The arrival of R1b1 carriers in Cameroon might be partly responsible for the Bantu expansion. R1b1 is found at very low frequencies in Bantu populations in southwestern Africa, and Cameroon is where the Bantu expansion began, largely due to their knowledge of agriculture.
R1b1 populations seems to have expanded throughout Africa from one population that lived along the Nile River. They arrived in the region from Western Asia bringing new technologies and the European LP SNP to the region. The predominately E1b1a Bantus adopted agriculture from the R1b1 guys that brought the technology to Africa from Western Asia.
However, the presence of lineages belonging to haplogroup R1b1* might represent footprints of demographic expansions in Central Africa not directly related to the “Bantu expansion.”
Haplogroup R or R-M207 is a Y-chromosome DNA haplogroup. It is both numerous and widespread amongst modern populations. Some descendant subclades are common throughout Europe, Central Asia and South Asia, and also common in parts of the West Asia, Africa and North America. Others are primarily from West Asia and South Asia.
The SNP defining this haplogroup is believed to have arisen during the Upper Paleolithic era: about 19,000 – 34,000 years ago. The most likely geographical location for this is Central Asia.
Haplogroup R* originated in North Asia just before the Last Glacial Maximum (26,500-19,000 years ago). The sole example of basal haplogroup R* has been found in the remains of a boy, known as MA-1, who lived at Mal’ta, an archaeological culture of the Upper Paleolithic (c. 24,000 to 15,000 BP) on the upper Angara River in the area west of Lake Baikal in the Irkutsk Oblast, Siberia, Russian Federation, 24,000 years ago.
Research published in 2014 suggests that a Mal’ta like people were important genetic contributors to the American Indians, Europeans, and central and south Asians but did not contribute to and was not related to East Eurasians.
The suggested similarity between Mal’ta and Upper Paleolithic civilizations of Western and Eastern Europe coincides with a long-held belief that the ancient people of Mal’ta were related to the Paleolithic societies of Europe.
These similarities can be established by their tools, dwelling structures, and art. These commonalities draw into question the origin of Upper Paleolithic Siberian people, and whether the migrating peoples originated from Southeastern Asia or quite possibly from Europe.
As a paper by Pille Hallast, Chiara Batini, Daniel Zadik and others put in 2014, the most recent common ancestor of all members of haplogroup R lived 19,000 years ago, “both R1a and R1b comprise young, star-like expansions” and present day Central Asian samples belong to “the deepest subclade of R1b-M269, while another, in a Bhutanese individual, forms an outgroup almost as old as the R1a/R1b split.”
Y-haplogroup R-M207 is common throughout Europe, South Asia and Central Asia. It also occurs in the Caucasus and Siberia. Some minorities in Africa also carry subclades of R-M207 at high frequencies.
While some indigenous peoples of the Americas and Australasia also feature high levels of R-M207, it is unclear whether these are deep-rooted, or an effect of European colonisation during the early modern era.
Autosomally, this Paleolithic population appears to have contributed mostly to the ancestry of modern Europeans and South Asians, the two regions where haplogroup R also happens to be the most common nowadays (R1b in Western Europe, R1a in Eastern Europe, Central and South Asia, and R2 in South Asia).
The oldest forms of R1b (M343, P25, L389) are found dispersed at very low frequencies from Western Europe to India, a vast region where could have roamed the nomadic R1b hunter-gatherers during the Ice Age.
The early R1b cattle herders would have split in at least three groups. The three main branches of R1b1 (R1b1a, R1b1b, R1b1c) all seem to have stemmed from the Middle East.
It has been hypothetised that R1b people (perhaps alongside neighbouring J2 tribes) were the first to domesticate cattlein northern Mesopotamia some 10,500 years ago. R1b tribes descended from mammoth hunters, and when mammoths went extinct, they started hunting other large game such as bisons and aurochs.
With the increase of the human population in the Fertile Crescent from the beginning of the Neolithic (starting 12,000 years ago), selective hunting and culling of herds started replacing indiscriminate killing of wild animals.
Haplogroup J2 is thought to have appeared somewhere in the Middle East towards the end of the last glaciation, between 15,000 and 22,000 years ago. The oldest known J2 sample at present comes from Kotias Klde in Georgia and dates from c. 9700 BCE, confirming that haplogroup J2 was already found around the Caucasus during the Mesolithic period.
Its present geographic distribution argues in favour of a Neolithic expansion from the Fertile Crescent. This expansion probably correlated with the diffusion of domesticated of cattle and goats (starting c. 8000-9000 BCE) from the Zagros mountains and northern Mesopotamia, rather than with the development of cereal agriculture in the Levant (which appears to be linked rather to haplogroups G2 and E1b1b).
A second expansion of J2 could have occured with the advent of metallurgy, notably copper working (from the Lower Danube valley, central Anatolia and northern Mesopotamia), and the rise of some of the oldest civilizations.
Quite a few ancient Mediterranean and Middle Eastern civilizations flourished in territories where J2 lineages were preponderant. This is the case of the Hattians, the Hurrians, the Etruscans, the Minoans, the Greeks, the Phoenicians (and their Carthaginian offshoot), the Israelites, and to a lower extent also the Romans, the Assyrians and the Persians. All the great seafaring civilizations from the middle Bronze Age to the Iron Age were dominated by J2 men.
There is a distinct association of ancient J2 civilizations with bull worship. The oldest evidence of a cult of the bull can be traced back to Neolithic central Anatolia, notably at the sites of Çatalhöyük and Alaca Höyük. Bull depictions are omnipresent in Minoan frescos and ceramics in Crete. Bull-masked terracotta figurines and bull-horned stone altars have been found in Cyprus (dating back as far as the Neolithic, the first presumed expansion of J2 from West Asia).
The Hattians, Sumerians, Babylonians, Canaaites, and Carthaginians all had bull deities (in contrast with Indo-European or East Asian religions). The sacred bull of Hinduism, Nandi, present in all temples dedicated to Shiva or Parvati, does not have an Indo-European origin, but can be traced back to Indus Valley civilization.
Minoan Crete, Hittite Anatolia, the Levant, Bactria and the Indus Valley also shared a tradition of bull leaping, the ritual of dodging the charge of a bull. It survives today in the traditional bullfighting of Andalusia in Spain and Provence in France, two regions with a high percentage of J2 lineages.
The increased involvement of humans in the life of aurochs, wild boars and goats led to their progressive taming. Cattle herders probably maintained a nomadic or semi-nomadic existence, while other people in the Fertile Crescent settled down to cultivate the land or keep smaller domesticates.
The analysis of bovine DNA has revealed that all the taurine cattle (Bos taurus) alive today descend from a population of only 80 aurochs. The earliest evidence of cattle domestication dates from circa 8,500 BCE in the Pre-Pottery Neolithic cultures in the Taurus Mountains.
The two oldest archaeological sites showing signs of cattle domestication are the villages of Çayönü Tepesi in southeastern Turkey and Dja’de el-Mughara in northern Iraq, two sites only 250 km away from each others. This is presumably the area from which R1b lineages started expanding – or in other words the “original homeland” of R1b.
R1b1b (M335), who by judging from its extreme rarity today wasn’t very successful, perhaps due to the heavy competition with other Neolithic populations in Anatolia, or to the scarcity of pastures in this mountainous environment, has only been found in Anatolia.
The southern branch, R1b1c (V88), is found mostly in the Levant and Africa. It migrated south to the Levant, where it became the V88 branch. Some of them searched for new lands south in Africa, first in Egypt, then colonising most of northern Africa, from the Mediterranean coast to the Sahel.
The northern branch, R1b1a (P297), seem to have originated around the Caucasus, eastern Anatolia or northern Mesopotamia, then to have crossed over the Caucasus, from where they would have invaded Europe and Central Asia. It crossed the Caucasus into the vast Pontic-Caspian Steppe, which provided ideal grazing grounds for cattle.
They split into two factions: R1b1a1 (M73), which went east along the Caspian Sea to Central Asia, and R1b1a2 (M269), which at first remained in the North Caucasus and the Pontic Steppe between the Dnieper and the Volga.
It is not yet clear whether M73 actually migrated across the Caucasus and reached Central Asia via Kazakhstan, or if it went south through Iran and Turkmenistan. In the latter case, M73 might not be an Indo-European branch of R1b, just like V88 and M335.
R1b-M269 (the most common form in Europe) is closely associated with the diffusion of Indo-European languages, as attested by its presence in all regions of the world where Indo-European languages were spoken in ancient times, from the Atlantic coast of Europe to the Indian subcontinent. The history of R1b and R1a are intricately connected to each others.
Like its northern counterpart (R1b-M269), R1b-V88 is associated with the domestication of cattle in northern Mesopotamia. Both branches of R1b probably split soon after cattle were domesticated, approximately 10,500 years ago (8,500 BCE).
R1b-V88 migrated south towards the Levant and Egypt. The migration of R1b people can be followed archeologically through the presence of domesticated cattle, which appear in central Syria around 8,000-7,500 BCE (late Mureybet period), then in the Southern Levant and Egypt around 7,000-6,500 BCE (e.g. at Nabta Playa and Bir Kiseiba).
Cattle herders subsequently spread across most of northern and eastern Africa. The Sahara desert would have been more humid during the Neolithic Subpluvial period (c. 7250-3250 BCE), and would have been a vast savannah full of grass, an ideal environment for cattle herding.
Evidence of cow herding during the Neolithic has shown up at Uan Muhuggiag in central Libya around 5500 BCE, at the Capeletti Cave in northern Algeria around 4500 BCE. But the most compelling evidence that R1b people related to modern Europeans once roamed the Sahara is to be found at Tassili n’Ajjer in southern Algeria, a site famous pyroglyphs (rock art) dating from the Neolithic era. Some painting dating from around 3000 BCE depict fair-skinned and blond or auburn haired women riding on cows.
After reaching the Maghreb, R1b-V88 cattle herders could have crossed the Strait of Gibraltar to Iberia, probably accompanied by G2 farmers, J1 and T1a goat herders and native Maghreban E-M81 lineages. These Maghreban Neolithic farmers/herders could have been the ones who established the Almagra Pottery culture in Andalusia in the 6th millennium BCE.
Nowadays small percentages (1 to 4%) of R1b-V88 are found in the Levant, among the Lebanese, the Druze, and the Jews, and almost in every country in Africa north of the equator. Higher frequency in Egypt (5%), among Berbers from the Egypt-Libya border (23%), among the Sudanese Copts (15%), the Hausa people of Sudan (40%), the the Fulani people of the Sahel (54% in Niger and Cameroon), and Chadic tribes of northern Nigeria and northern Cameroon (especially among the Kirdi), where it is observed at a frequency ranging from 30% to 95% of men.
According toCruciani et al. (2010) R1b-V88 would have crossed the Sahara between 9,200 and 5,600 years ago, and is most probably associated with the diffusion of Chadic languages, a branch of the Afroasiatic languages. V88 would have migrated from Egypt to Sudan then expanded along the Sahel until northern Cameroon and Nigeria. However, R1b-V88 is not only present among Chadic speakers, but also among Senegambian speakers (Fula-Hausa) and Semitic speakers (Berbers, Arabs).
R1b-V88 is found among the native populations of Rwanda, South Africa, Namibia, Angola, Congo, Gabon, Equatorial Guinea, Ivory Coast, Guinea-Bissau. The wide distribution of V88 in all parts of Africa, its incidence among herding tribes, and the coalescence age of the haplogroup all support a Neolithic dispersal. In any case, a later migration out of Egypt would be improbable since it would have brought haplogroups that came to Egypt during the Bronze Age, such as J1, J2, R1a or R1b-L23.
Afroasiatic (Afro-Asiatic), also known as Afrasian and traditionally as Hamito-Semitic (Chamito-Semitic), is a large language family of several hundred related languages and dialects. It includes languages spoken predominantly in the Middle East, North Africa, the Horn of Africa, and parts of the Sahel. It has six branches: Berber, Chadic, Cushitic, Egyptian, Omotic and Semitic.
The term Afroasiatic Urheimat (Urheimat meaning “original homeland” in German) refers to the ‘hypothetical’ place where Proto-Afroasiatic speakers lived in a single linguistic community, or complex of communities, before this original language dispersed geographically and divided into distinct languages.
There is no agreement when or where the original homeland of this language family existed. Proposed locations include North Africa, the Horn of Africa, the Eastern Sahara, and the Levant. Their distribution seems to have been influenced by the Saharan pump operating over the last 10,000 years. Afro-Asiatic languages are today primarily spoken in the Middle East, North Africa, the Horn of Africa, and parts of the Sahel.
Estimates of the date at which the Proto-Afroasiatic language was spoken vary widely. They fall within a range between approximately 7,500 BC (9,500 years ago) and approximately 16,000 BC (18,000 years ago).
According to Igor M. Diakonoff (1988: 33n), Proto-Afroasiatic was spoken c. 10,000 BC. According to Christopher Ehret (2002: 35–36), Proto-Afroasiatic was spoken c. 11,000 BC at the latest, and possibly as early as c. 16,000 BC. These dates are older than dates associated with most other proto-languages.
The Chadic languages are a branch of the Afroasiatic language family that is spoken in parts of the Sahel. They include 150 languages spoken across northern Nigeria, southern Niger, southern Chad, Central African Republic and northern Cameroon. The most widely spoken Chadic language is Hausa, a lingua franca of much of inland West Africa.
Several modern genetic studies of Chadic speaking groups in the northern Cameroon region have observed high frequencies of the Y-Chromosome Haplogroup R1b in these populations (specifically, of R1b’s R-V88 variant). This paternal marker is common in parts of West Eurasia, but otherwise rare in Africa. Cruciani et al. (2010) thus propose that the Proto-Chadic speakers during the mid-Holocene (~7,000 years ago) migrated from the Levant to the Central Sahara, and from there settled in the Lake Chad Basin.
The Nok culture appeared in Northern Nigeria around 1000 BC and vanished under unknown circumstances around 300 AD in the region of West Africa. It is believed to be the product of an ancestral nation that branched to create the Hausa, Gwari, Birom, Kanuri, Nupe and Jukun peoples. The Kwatarkwashi Culture or Sokoto Culture located to the North west of Nok is thought to be the same as or an earlier ancestor of the Nok.
Nok’s social system is thought to have been highly advanced. The Nok culture is considered to be the earliest sub-Saharan producer of life-sized Terracotta. Images show figures on horseback, indicating that the Nok culture possessed the horse. Iron use, in smelting and forging for tools, appears in Nok culture in Africa at least by 550 BC and possibly earlier.
Two major sources of information are used to determine where and when the horse was first domesticated and how the domesticated horse spread around the world. The first source is based on palaeological and archeological discoveries; the second source is a comparison of DNA obtained from modern horses to that from bones and teeth of ancient horse remains.
The earliest archaeological evidence for the domestication of the horse comes from sites in Ukraine and Kazakhstan, dating to approximately 3500–4000 BC. By 3000 BC, the horse was completely domesticated and by 2000 BC there was a sharp increase in the number of horse bones found in human settlements in northwestern Europe, indicating the spread of domesticated horses throughout the continent.
The most recent, but most irrefutable evidence of domestication comes from sites where horse remains were interred with chariots in graves of the Sintashta and Petrovska cultures c. 2100 BC.
Domestication is also studied by using the genetic material of present-day horses and comparing it with the genetic material present in the bones and teeth of horse remains found in archaeological and palaeological excavations.
The variation in the genetic material shows that very few wild stallions contributed to the domestic horse, while many mares were part of early domesticated herds. This is reflected in the difference in genetic variation between the DNA that is passed on along the paternal, or sire line (Y-DNA) versus that passed on along the maternal, or dam line (mtDNA).
There are very low levels of Y-chromosome variability, but a great deal of genetic variation in mitochondrial DNA. There is also regional variation in mitochondrial DNA due to the inclusion of wild mares in domestic herds.
Another characteristic of domestication is an increase in coat color variation. In horses, this increased dramatically between 5000 and 3000 BC.
Bantu peoples is used as a general label for the 300–600 ethnic groups in Africa who speak Bantu languages. They inhabit a geographical area stretching east and southward from Central Africa across the African Great Lakes region down to Southern Africa. Bantu is a major branch of the Niger-Congo language family spoken by most populations in Africa.
Proto-Bantu (also Common Bantu) is the reconstructed common ancestor of most Bantu languages. It is thought to have originally been spoken in West/Central Africa in the area of what is now Cameroon. Approximately 3000–4000 years ago, it split off from other Niger-Congo languages when the Bantu people began their migration to the south and east.
The word Bantu, and its variations, means “people” or “humans”. The root in Proto-Bantu is reconstructed as *-ntu. Versions of the word Bantu (that is, the root plus the class 2 noun class prefix *ba-) occur in all Bantu languages.
E-M243 (especially its subclades M78 and M81) is found at high frequencies in North East Africa and North Africa and is the only subclade that is found in Europe and Asia at significant frequencies.
E-M243 is common among Afro-Asiatic speakers in the Near East and North Africa as well as among some Nilo-Saharan and Niger–Congo speakers in North East Africa and Sudan. E-M243 is far less common in West, Central, and Southern Africa.
Haplogroup E-V38 has two basal branches, E-M329 (formerly E1b1c) and E-M2 (formerly E1b1a), the former is almost exclusively found in Ethiopia. The E-M2 branches are the predominant lineage in Western Africa, Central Africa, Southern Africa, and the southern parts of Eastern Africa.
The discovery of two SNPs (V38 and V100) by Trombetta et al. (2011) significantly redefined the E-V38 phylogenetic tree. This led the authors to suggests that E-V38 may have originated in East Africa. V38 joins the West African-affiliated E-M2 and the northern East African-affiliated E-M329 with an earlier common ancestor who, like E-P2, may have also originated in East Africa.
It is possible that soon after the evolution of E-V38, trans-Saharan migrants carried the E-V38 marker to Northern Africa and Central Africa and/or West Africa where the more common E-M2 marker later arose and became prolific within the last 20,000-30,000 years.
The downstreams SNP E-M180 possibly originated on the moist south-central Saharan savannah/grassland of northern West Africa during the early Holocene period. Much of the population that carried E-M2 retreated to southern West Africa with the drying of the Sahara. These later people migrated from Southeastern Nigeria and Cameroon ~8.0 kya to Central Africa, East Africa, and Southern Africa causing or following the Bantu expansion.
According to Wood et al. (2005) and Rosa et al. (2007), such population movements from West Africa changed the pre-existing population Y chromosomal diversity in Western, Central, Southern and southern East Africa, replacing the previous haplogroups frequencies in these areas with the now dominant E1b1a1 lineages.
Traces of earlier inhabitants, however, can be observed today in these regions via the presence of the Y DNA haplogroups A1a, A1b, A2, A3, and B-M60 that are common in certain populations, such as the Mbuti and Khoisan.
E-V38 reaches, the most prevalent subclade of E in Africa, frequencies of over 80% in many parts of West Africa, Central Africa, East Africa as well as Southern Africa. It is observed at high frequencies in all African regions except the northernmost and easternmost portions of the continent.
This haplogroup’s frequency and diversity are highest in the West Africa region. Within Africa, E-V38 displays a west-to-east as well as a south-to-north clinal distribution. In other words, the frequency of the haplogroup decreases as one move from western and southern Africa toward the eastern and northern parts of the continent.
About 3000 years ago, speakers of the proto-Bantu language group began a millennia-long series of migrations eastward from their homeland between West Africa and Central Africa at the border of eastern Nigeria and Cameroon.
This Bantu expansion first introduced Bantu peoples to central, southern, and southeastern Africa, regions they had previously been absent from. The proto-Bantu migrants in the process assimilated and/or displaced a number of earlier inhabitants that they came across, including Khoisan populations in the south and Afro-Asiatic groups in the southeast.
The linguistic core of the Bantu family of languages, a branch of the Niger–Congo language family, was located in the adjoining region of Cameroon and Nigeria. From this core, expansion began about 3,000 years ago, with one stream going into East Africa, and other streams going south along the African coast of Gabon, Democratic Republic of the Congo, and Angola, or inland along the many south to north flowing rivers of the Congo River system. The expansion eventually reached South Africa probably as early as AD 300.
The hypothesized Bantu expansion pushed out or assimilated the hunter-forager proto-Khoisan, who formerly inhabited Southern Africa. In Eastern and Southern Africa, Bantu-speakers may have adopted livestock husbandry from other unrelated Cushitic- and Nilotic-speaking peoples they encountered.
Herding practices reached the far south several centuries before Bantu-speaking migrants did. Archaeological, linguistic, genetic, and environmental evidence all support the conclusion that the Bantu expansion was a significant human migration. It is thought that Central African Pygmies and Bantus branched out from a common ancestral population c. 70,000 years ago.
Current scholarly understanding places the ancestral proto-Bantu homeland in West Africa near the present-day southwestern border of Nigeria and Cameroon c. 4,000 years ago (2000 B.C.), and regards the Bantu languages as a branch of the Niger–Congo language family.
This view represents a resolution of debates in the 1960s over competing theories advanced by Joseph Greenberg and Malcolm Guthrie, in favor of refinements of Greenberg’s theory.
It seems likely that the expansion of the Bantu-speaking people from their core region in West Africa began around 1000 BCE. Although early models posited that the early speakers were both iron-using and agricultural, archaeology has shown that they did not use iron until as late as 400 BCE, though they were agricultural.
Based on wide comparisons including non-Bantu languages, Greenberg argued that Proto-Bantu, the hypothetical ancestor of the Bantu languages, had strong ancestral affinities with a group of languages spoken in Southeastern Nigeria. He proposed that Bantu languages had spread east and south from there, to secondary centers of further dispersion, over hundreds of years.
Before the expansion of farming and herding peoples, including those speaking Bantu languages, Africa south of the equator was populated by neolithic hunting and foraging peoples. Some of them were ancestral to proto-Khoisan-speaking peoples, whose modern hunter-forager and linguistic descendants, the Khoekhoe and San, occupy the arid regions around the Kalahari desert. The Hadza and Sandawe populations in Tanzania comprise the other modern hunter-forager remnant in Africa of these proto-Khoisan-speaking peoples.
The western branch, not necessarily linguistically distinct, according to Christopher Ehret, followed the coast and the major rivers of the Congo system southward, reaching central Angola by around 500 BCE.
It is clear that there were human populations in the region at the time of the expansion, and Pygmies are their purer descendants. However, mtDNA genetic research from Cabinda suggests that only haplogroups that originated in West Africa are found there today, and the distinctive L0 of the pre-Bantu population is missing, suggesting that there was a complete population replacement. In South Africa, however, a more complex intermixing could have taken place.
Further east, Bantu-speaking communities had reached the great Central African rainforest, and by 500 BCE, pioneering groups had emerged into the savannas to the south, in what are now the Democratic Republic of Congo, Angola, and Zambia.
Another stream of migration, moving east by 3,000 years ago (1000 BCE), was creating a major new population center near the Great Lakes of East Africa, where a rich environment supported a dense population. Movements by small groups to the southeast from the Great Lakes region were more rapid, with initial settlements widely dispersed near the coast and near rivers, due to comparatively harsh farming conditions in areas further from water.
Pioneering groups had reached modern KwaZulu-Natal in South Africa by CE 300 along the coast, and the modern Limpopo Province (formerly Northern Transvaal) by CE 500.
Over a period of many centuries, most hunting-foraging peoples were displaced and absorbed by incoming Bantu-speaking communities, as well as by Ubangian, Nilotic, and Sudanic language-speakers in North Central and Eastern Africa.
The Bantu expansion was a long series of physical migrations, a diffusion of language and knowledge out into and in from neighboring populations, and a creation of new societal groups involving inter-marriage among communities and small groups moving to communities and small groups moving to new areas.
After their movements from their original homeland in West Africa, Bantus also encountered in East Africa peoples of Afro-Asiatic (mainly Cushitic) and Nilo-Saharan (mainly Nilotic and Sudanic) ancestral origin. As cattle terminology in use amongst the few modern Bantu pastoralist groups suggests, the Bantu migrants would acquire cattle from their new Cushitic neighbors.
Geneticist and anthropologists previously suspected that ancient Africans domesticated cattle native to the African continent nearly 10,000 years ago. Now, a team of University of Missouri researchers has completed the genetic history of 134 cattle breeds from around the world. In the process of completing this history, they found that ancient domesticated African cattle originated in the “Fertile Crescent,” a region that covered modern day Iraq, Jordan, Syria and Israel.
Lead researcher Jared Decker, an assistant professor of animal science in the MU College of Agriculture, Food and Natural Resources, says the genetics of these African cattle breeds are similar to those of cattle first domesticated in the Middle East nearly 10,000 years ago, proving that those cattle were brought to Africa as farmers migrated south. Those cattle then interbred with wild cattle, or aurochs, which were native to the region, and changed their genetic makeup enough to confuse geneticists.
“In many ways, the history of cattle genetics mirrors human history,” Decker said. “In the case of African cattle, anthropologists and geneticists used to suspect that domesticated African cattle were native to the continent, when in fact, they were brought by migrating peoples thousands of years ago. By better understanding the history of the animals we domesticate, we can better understand ourselves.”
Linguistic evidence also indicates that Bantus likely borrowed the custom of milking cattle directly from Cushitic peoples in the area. Later interactions between Bantu and Cushitic peoples resulted in Bantu groups with significant Cushitic ethnic admixture, such as the Tutsi of the African Great Lakes region; and culturo-linguistic influences, such as the Herero herdsmen of southern Africa.
On the coastal section of East Africa, another mixed Bantu community developed through contact with Muslim Arab and Persian traders. The Swahili culture that emerged from these exchanges evinces many Arab and Islamic influences not seen in traditional Bantu culture, as do the many Afro-Arab members of the Bantu Swahili people.
“Khoisan” (also spelled Khoesaan, Khoesan or Khoe–San) is a unifying name for two groups of peoples of Southern Africa, who share physical and putative linguistic characteristics distinct from the Bantu majority of the region. Culturally, the Khoisan are divided into the foraging San, or Bushmen, and the pastoral Khoi, or more specifically Khoikhoi, previously known as Hottentots.
The San include the indigenous inhabitants of Southern Africa before the southward Bantu migrations from Central and East Africa reached their region, which led to the Bantu populations displacing the Khoi and San to become the predominant inhabitants of Southern Africa. The distinct origin of the Khoi is debated.
Over time, some Khoi abandoned pastoralism and adopted the hunter-gatherer economy of the San, probably due to a drying climate, and are now considered San. Similarly, the Bantu Damara people who migrated south later abandoned agriculture and adopted the Khoi economy. Large Khoisan populations remain in several arid areas in the region, notably in the Kalahari Desert.
From the beginning of the Upper Paleolithic periods, hunting and gathering cultures known as the Sangoan occupied southern Africa in areas where annual rainfall is less than a metre (1000 mm; 40 in), and today’s San and Khoi people resemble the ancient Sangoan skeletal remains.
These people in parts of southern Africa were the ancestors of the Khoisan people who inhabited the Kalahari Desert. A set of tools almost identical to that used by the modern San and dating to 44,000 BCE was discovered at Border Cave in KwaZulu-Natal in 2012.
Probably due to their region’s lack of suitable candidates for domestication, the Khoisan did not have farming or domesticated chickens until a few hundred years ago, when they adopted the domesticated cattle and sheep of the Bantu that had spread in advance of the people’s actual arrival.
Against the traditional interpretation that finds a common origin for the Khoi and San, other evidence has suggested that the ancestors of the Khoi peoples (one subset of the Khoisan) are relatively recent pre-Bantu agricultural immigrants to southern Africa, who abandoned agriculture as the climate dried and either joined the San as hunter-gatherers or retained pastoralism to become the Khoikhoi.
In the 1990s, genomic studies of worlds peoples found that the Y chromosome of San men share certain patterns of polymorphisms that are distinct from those of all other populations.
Because the Y chromosome is highly conserved between generations, this type of DNA test is used to determine when different subgroups separated from one another, and hence their last common ancestry.
The authors of these studies suggested that the San may have been one of the first populations to differentiate from the most recent common paternal ancestor of all extant humans, the so-called Y-chromosomal Adam by patrilineal descent, estimated to have lived 60,000 to 90,000 years ago.
The authors also note that their results should be interpreted as only finding that the Khoisan “preserve ancient lineages”, and not that they “stopped evolving” or are an “ancient group”, since subsequent changes in their population are in parallel and similar to those of all other human populations.
Various Y-chromosome studies since confirmed that the Khoisan carry some of the most divergent (oldest) Y-chromosome haplogroups. These haplogroups are specific sub-groups of haplogroups A and B, the two earliest branches on the human Y-chromosome tree.
Similar to findings from Y-Chromosome studies, mitochondrial DNA studies also showed evidence that the Khoe–San people carry high frequencies of the earliest haplogroup branches in the human mitochondrial DNA tree.
The most divergent (oldest) mitochondrial haplogroup, L0d, have been identified at its highest frequencies in the southern African Khoe and San groups. The distinctiveness of the Khoisan in both matrilineal and patrilineal groupings is a further indicator that they represent a population historically distinct from other Africans.
In modern South Africa and Namibia, a large share of the Cape Coloureds, Xhosa and Tswana is of partial Khoisan descent, by far outnumbering the unassimilated Khoisan population.
Various Y chromosome studies show that the San carry some of the most divergent (oldest) human Y-chromosome haplogroups. These haplogroups are specific sub-groups of haplogroups A and B, the two earliest branches on the human Y-chromosome tree.
Mitochondrial DNA studies also provide evidence that the San carry high frequencies of the earliest haplogroup branches in the human mitochondrial DNA tree. This DNA is inherited only from one’s mother.
In a study published in March 2011, Brenna Henn and colleagues found that the ǂKhomani San, as well as the Sandawe and Hadza peoples of Tanzania, were the most genetically diverse of any living humans studied. This high degree of genetic diversity hints at the origin of anatomically modern humans.
The Khoikhoi, originally part of a pastoral culture and language group to be found across Southern Africa, originated in the northern area of modern Botswana. The Khoikhoi tribe, described as the Hottentots, had occupied the behind belt for centuries before the Dutch arrived. Southward migration of the ethnic group was steady, eventually reaching the Cape approximately 2,000 years ago.
The genus Homo evolved and diverged from other hominins in Africa, after the human clade split from the chimpanzee lineage of the hominids (great apes) branch of the primates.
According to the Sahara pump theory evidence suggests that genus Homo have migrated out of Africa at least three and possibly four times (e.g. Homo erectus, Homo heidelbergensis and two or three times for Homo sapiens).
Modern humans (Homo sapiens, primarily ssp. Homo sapiens sapiens) are the only extant members of Hominina clade (or human clade), a branch of the taxonomical tribe Hominini belonging to the family of great apes.
They are characterized by erect posture and bipedal locomotion; manual dexterity and increased tool use, compared to other animals; and a general trend toward larger, more complex brains and societies.
Early hominins—particularly the australopithecines, whose brains and anatomy are in many ways more similar to ancestral non-human apes—are less often referred to as “human” than hominins of the genus Homo.
Like other early humans that were living at this time, they gathered and hunted food, and evolved behaviors that helped them respond to the challenges of survival in unstable environments.
Several of these hominins used fire, occupied much of Eurasia, and gave rise to anatomically modern Homo sapiens in Africa about 200,000 years ago – the earliest remains from that time were uncovered near the Omo River in Ethiopia.
They began to exhibit evidence of behavioral modernity around 50,000 years ago. In several waves of migration, anatomically modern humans ventured out of Africa and populated most of the world.
The entire human race outside Africa owes its existence to the survival of a single tribe of around 200 people who crossed the Red Sea 70,000 years ago, scientists have discovered.
This dispersal out of Africa is estimated to have begun about 70,000 years BP from Northeast Africa. Current evidence suggests that there was only one such dispersal and that it only involved a few hundred individuals. The vast majority of humans stayed in Africa and adapted to a diverse array of environments.
Modern humans subsequently spread globally, replacing earlier hominins (either through competition or hybridization). They inhabited Eurasia and Oceania by 40,000 years BP, and the Americas at least 14,500 years BP.
Modern humans, defined as the species Homo sapiens or specifically to the single extant subspecies Homo sapiens sapiens, proceeded to colonize all the continents and larger islands, arriving in Eurasia 125,000–60,000 years ago, Australia around 40,000 years ago, the Americas around 15,000 years ago, and remote islands such as Hawaii, Easter Island, Madagascar, and New Zealand between the years 300 and 1280.
Though most of human existence has been sustained by hunting and gathering in band societies, increasing numbers of human societies began to practice sedentary agriculture approximately some 10,000 years ago, domesticating plants and animals, thus allowing for the growth of civilization.
The advent of agriculture prompted the Neolithic Revolution, when access to food surplus led to the formation of permanent human settlements, the domestication of animals and the use of metal tools for the first time in history. Agriculture encouraged trade and cooperation, and led to complex society. Because of the significance of this date for human society, it is the epoch of the Holocene calendar or Human Era.
“Khoisan” is a unifying name for two groups of peoples of Southern Africa, who share physical and putative linguistic characteristics distinct from the Bantu majority of the region. Culturally, the Khoisan are divided into the foraging San, or Bushmen, and the pastoral Khoi, or more specifically Khoikhoi, previously known as Hottentots.
In the 1990s, genomic studies of worlds peoples found that the Y chromosome of San men share certain patterns of polymorphisms that are distinct from those of all other populations.
Because the Y chromosome is highly conserved between generations, this type of DNA test is used to determine when different subgroups separated from one another, and hence their last common ancestry.
The authors of these studies suggested that the San may have been one of the first populations to differentiate from the most recent common paternal ancestor of all extant humans, the so-called Y-chromosomal Adam by patrilineal descent, estimated to have lived 60,000 to 90,000 years ago.
The authors also note that their results should be interpreted as only finding that the Khoisan “preserve ancient lineages”, and not that they “stopped evolving” or are an “ancient group”, since subsequent changes in their population are in parallel and similar to those of all other human populations.
In human genetics, Mitochondrial Eve is the matrilineal most recent common ancestor (MRCA) of all currently living humans. This is the most recent woman from whom all living humans today descend, in an unbroken line, on their mother’s side, and through the mothers of those mothers, and so on, back until all lines converge on one woman, who is estimated to have lived approximately 100,000–200,000 years ago.
Because all mitochondrial DNA (mtDNA) generally (but see paternal mtDNA transmission) is passed from mother to offspring without recombination, all mtDNA in every living person is directly descended from hers by definition, differing only by the mutations that over generations have occurred in the germ cell mtDNA since the conception of the original “Mitochondrial Eve”.
Mitochondrial Eve is named after mitochondria and the biblical Eve. Unlike her biblical namesake, she was not the only living human female of her time. However, her female contemporaries, excluding her mother, failed to produce a direct unbroken female line to any living person in the present day.
Mitochondrial Eve is estimated to have lived between 99,000 and 200,000 years ago, most likely in East Africa, when Homo sapiens sapiens (anatomically modern humans) were developing as a population distinct from other human sub-species.


Leave a comment